Strategic importance of orthodontic buccal tubes
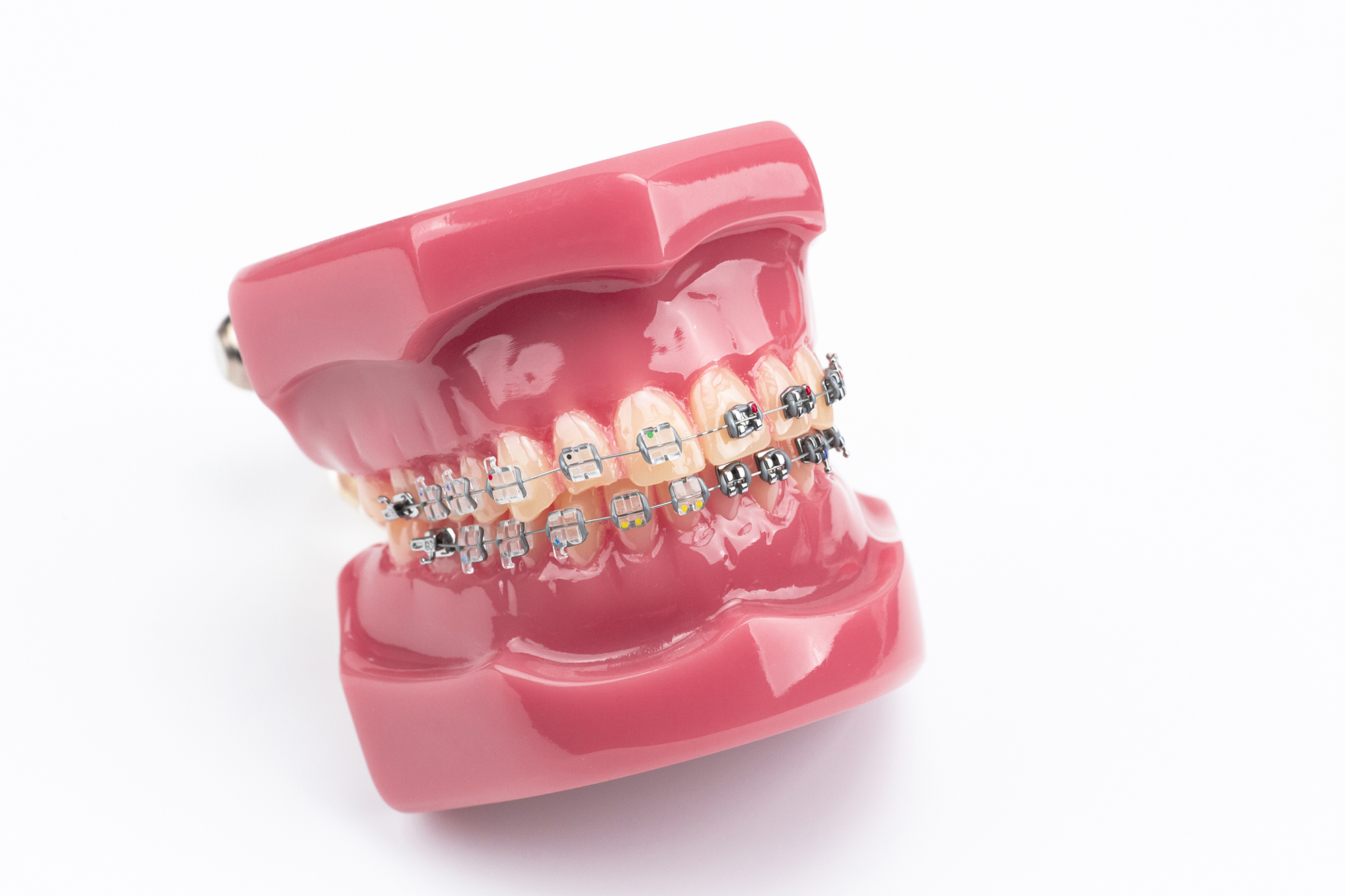
Orthodontic buccal tubes serve as foundational anchoring components in fixed orthodontic appliance systems. Positioned primarily on the first and second molars, these precision-engineered receptacles secure the ends of archwires, transmitting the critical mechanical forces required for tooth movement, rotation, and torque. For B2B distributors and dental procurement teams, securing a reliable supply of high-quality buccal tubes is essential to maintaining a competitive edge in the dental consumables market.
Unlike brackets used on anterior teeth, buccal tubes endure the highest occlusal forces in the oral cavity. Consequently, their structural integrity directly impacts the clinical success of the entire orthodontic treatment. Procurement strategies must therefore balance rigorous metallurgical standards with favorable unit economics to support the operational needs of high-volume orthodontic clinics.
Category role in orthodontic product portfolios
Within a comprehensive orthodontic product portfolio, buccal tubes function as an indispensable anchor product. They are universally required in almost every traditional fixed braces treatment plan. Distributors typically position buccal tubes alongside complementary consumables such as self-ligating brackets, molar bands, nickel-titanium (NiTi) archwires, and elastomeric power chains. By maintaining a robust inventory of these tubes, wholesalers can offer complete “start-to-finish” clinical kits, increasing the average order value per clinical buyer.
Furthermore, the availability of diverse buccal tube prescriptions—such as Roth, MBT, and Edgewise—allows distributors to cater to various clinical philosophies. Offering both bondable tubes (which adhere directly to the enamel) and weldable tubes (which are pre-attached to molar bands) ensures comprehensive market coverage and reinforces the distributor’s role as a primary, single-source supplier.
Demand drivers and reorder patterns
Demand for buccal tubes is characterized by high volume and rapid turnover. Because they are single-use medical consumables, orthodontic clinics require a continuous, uninterrupted supply. Industry data indicates that established clinic-supplier relationships yield reorder rates exceeding 70%, making this category a highly predictable revenue stream for distributors.
Reorder patterns are closely tied to the academic calendar and regional demographic trends, with peak orthodontic starts often occurring during the summer months. Procurement managers must forecast inventory accordingly, ensuring that buffer stocks of standard 0.022-inch and 0.018-inch slot sizes are maintained to prevent stockouts during seasonal demand spikes.
Margin and commercial stability factors
Commercial stability in the buccal tube category is driven by a highly favorable value-to-weight ratio and strong gross margins. Wholesale acquisition costs for standard stainless steel buccal tubes typically range from $0.50 to $2.50 per unit, depending on manufacturing technology and order volumes. Regional distributors frequently achieve gross margins of 40% to 60% when selling to end-user clinics.
Margin protection relies heavily on minimizing clinical failure rates, specifically tube debonding and slot deformation. High-quality tubes that eliminate emergency clinical visits for replacements protect the supplier’s reputation and prevent margin erosion caused by returns and refunds. Consequently, investing in premium manufacturing techniques, such as Metal Injection Molding (MIM), acts as a direct safeguard for long-term commercial profitability.
Product specifications and design comparison
Evaluating the technical specifications of orthodontic buccal tubes is a critical step in the procurement process. Buyers must scrutinize metallurgical properties, base contour geometries, and slot tolerances to ensure the products meet the rigorous mechanical demands of modern orthodontic therapies. A thorough understanding of these design variables enables distributors to source products that offer superior clinical performance.
Core structural and material specifications
Modern high-tier buccal tubes are predominantly manufactured using 17-4 PH (Precipitation Hardening) stainless steel via Metal Injection Molding (MIM) technology. This material offers exceptional tensile strength, corrosion resistance, and biocompatibility. The MIM process allows for the creation of complex, low-friction geometries that are impossible to achieve with traditional casting or milling methods.
Standardization is critical in buccal tube specifications. The central slot must accommodate industry-standard archwire dimensions, universally offered in 0.018-inch or 0.022-inch heights. Precision tolerances of ±0.001 inches are required to ensure that the archwire expresses the correct torque and angulation without excessive play or binding.
Performance impact of key design variables
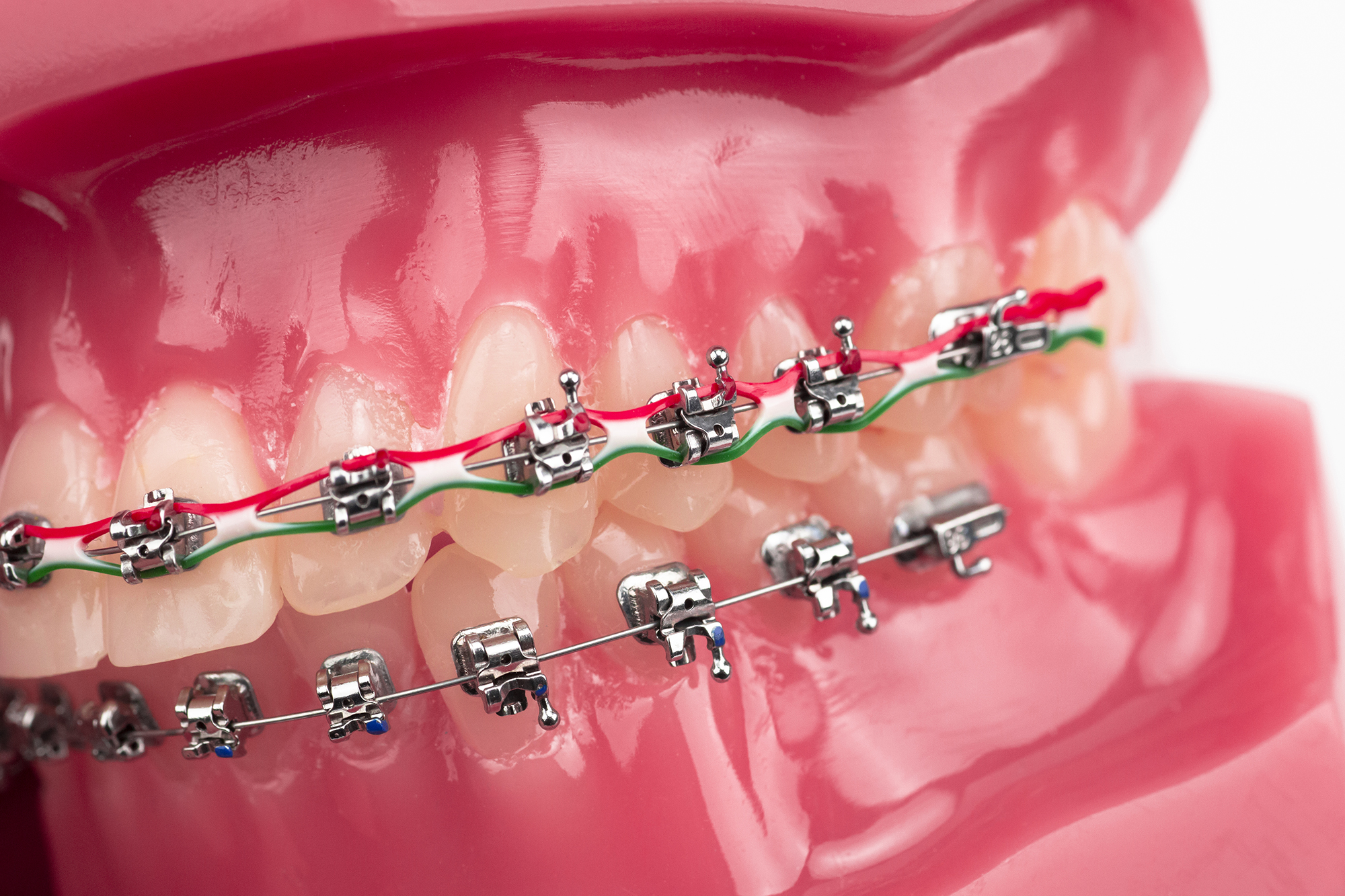
Key design variables significantly influence the clinical efficacy of buccal tubes. The anatomical contour of the tube’s base must closely match the natural curvature of the molar to ensure a uniform adhesive layer. Additionally, the inclusion of a trumpet-shaped or funneled mesial entrance drastically reduces the friction and chair time required for the clinician to insert the archwire.
Bond strength is heavily dictated by the base retention design. An 80-gauge woven mesh base or a micro-etched pylon base is a standard requirement. Clinical viability dictates that the shear bond strength must consistently exceed 10 MPa to withstand masticatory forces without premature debonding.
| Specification | Standard Requirement | Clinical Impact |
|---|---|---|
| Material | MIM 17-4 PH Stainless Steel | Prevents slot deformation under heavy wire torque |
| Slot Dimensions | 0.018″ or 0.022″ (±0.001″) | Ensures precise force expression and wire seating |
| Base Design | 80-gauge mesh or micro-pylon | Achieves >10 MPa shear bond strength to prevent debonding |
| Mesial Entrance | Funneled / Trumpet shape | Reduces wire insertion time and clinical friction |
Monoblock vs welded design trade-offs
Procurement teams frequently face the choice between monoblock and welded buccal tube designs. Welded designs feature a separate tube component that is laser-welded or brazed onto a distinct mesh pad. While this allows manufacturers to use larger, highly retentive mesh bases, it introduces a potential failure point at the weld seam, which can separate under extreme occlusal stress.
Conversely, monoblock buccal tubes are cast or injected as a single, unified piece of metal. This one-piece construction eliminates the risk of base-to-tube separation, resulting in a 0% detachment rate at the base junction. Monoblock designs also typically boast a lower vertical profile, which minimizes occlusal interference and improves patient comfort. For premium product lines, monoblock MIM construction is generally the preferred standard.
Supplier qualification and compliance review
Because buccal tubes are classified as medical devices, sourcing them requires rigorous vetting of supplier manufacturing capabilities and regulatory adherence. Distributors must partner with factories that implement stringent process controls to ensure batch-to-batch consistency and absolute clinical safety.
Factory capabilities and process controls
A qualified manufacturer of orthodontic consumables must demonstrate advanced production capabilities. Leading facilities utilize automated MIM production lines paired with high-precision German CNC equipment to maintain exact dimensional tolerances. High-capacity suppliers typically demonstrate output capabilities of 10,000 to 50,000 pieces weekly, ensuring they can scale alongside the distributor’s growth.
Process control must extend from raw material inspection to final polishing. Factories should employ advanced tumbling and electropolishing techniques to ensure smooth, rounded facial contours on the buccal tubes, preventing soft tissue irritation in the patient’s mouth. Buyers should request documentation of standard operating procedures (SOPs) governing furnace temperatures during the MIM sintering process, as this dictates the final density and strength of the steel.
Regulatory certifications and traceability
Regulatory compliance is a non-negotiable prerequisite for importing and distributing buccal tubes. For the European market, products must carry a CE mark and comply with the Medical Device Regulation (MDR). In the United States, manufacturers must hold an active FDA 510(k) clearance for orthodontic brackets and tubes.
Furthermore, the manufacturing facility must be certified under ISO 13485:2016, the internationally recognized quality management standard for medical devices. Traceability is equally critical; suppliers must implement Unique Device Identification (UDI) systems, allowing full backward traceability of lot numbers to the specific batch of raw 17-4 stainless steel used in production.
Supplier audit and quality verification steps
Conducting comprehensive supplier audits is vital before finalizing procurement contracts. Audits should verify the supplier’s adherence to ISO 10993 standards for the biological evaluation of medical devices, ensuring the alloys do not leach cytotoxic elements. Quality verification steps should include reviewing the factory’s Acceptable Quality Limit (AQL) protocols.
For high-precision items like buccal tubes, an AQL of 1.0 or 1.5 for major dimensional defects is standard. Buyers should request sample batches to conduct independent mechanical testing, specifically measuring slot height with pin gauges and executing shear bond strength tests on extracted teeth or analog materials to verify the manufacturer’s performance claims.
Purchasing, packaging, and logistics planning
Efficient supply chain execution determines the ultimate profitability of the buccal tube category. Procurement managers must carefully structure order volumes, define packaging specifications, and establish robust logistics protocols to minimize landed costs and prevent inventory disruptions.
MOQ, private label, and assortment decisions
Minimum Order Quantities (MOQs) for standard buccal tubes typically range from 1,000 to 5,000 sets (with a set comprising upper right, upper left, lower right, and lower left quadrants). Distributors looking to establish a private label brand must negotiate these MOQs alongside customization costs.
Private label strategies often include custom laser engraving on the base or facial surface of the tube for easy quadrant identification. This laser marking generally adds $0.10 to $0.20 per unit but significantly enhances the premium perception of the product. Assortment planning should heavily index toward single-tube configurations, though double and triple tubes should be stocked in lower quantities to accommodate complex extraction or surgical cases.
Packaging, sterilization, and labeling requirements
Packaging requirements for buccal tubes must balance clinical convenience with sterile barrier integrity. Products are typically packaged in patient-specific kits of four or in bulk clinical bags containing 10 to 50 pieces. Packaging materials must be highly durable and resistant to puncture by the sharp hooks of the tubes.
While buccal tubes are generally sold non-sterile and are sterilized chairside by the clinician via autoclaving, the packaging must clearly display critical regulatory information. This includes the manufacturer’s details, lot numbers, strict expiration dates (commonly rated for 5 years), and universal quadrant symbols (UR, UL, LR, LL) to prevent clinical placement errors.
Lead times, shipping, and defect management
Production lead times for bulk OEM buccal tube orders generally span 15 to 30 days, depending on the complexity of private label packaging and current factory capacity. Because buccal tubes possess an exceptionally high value-to-weight ratio, air freight is the standard logistics modality, ensuring rapid replenishment without incurring prohibitive shipping costs.
Defect management is a critical component of the logistics cycle. Distributors must negotiate clear Return Merchandise Authorization (RMA) protocols with the manufacturer. A standardized supplier agreement should stipulate full replacement or credit for any batch exhibiting a clinical debonding rate exceeding a mutually agreed threshold, typically set at 1% to 2% under normal clinical use.
Final sourcing and channel selection
The final phase of the procurement process involves defining the overarching channel strategy and selecting the ultimate sourcing model. Distributors must decide whether to act as authorized agents for established manufacturer brands or to cultivate their own proprietary OEM lines, a choice that fundamentally dictates business scalability.
Branded vs OEM channel strategy
Distributing branded buccal tubes from established manufacturers provides immediate market acceptance. Clinics are often loyal to specific brand names due to familiar clinical handling and proven track records. However, this strategy typically restricts the distributor to lower gross margins, generally hovering between 20% and 30%, and leaves them vulnerable to manufacturer price hikes or direct-to-consumer channel shifts.
Conversely, an OEM (Original Equipment Manufacturer) private label strategy allows the distributor to build unparalleled brand equity and command gross margins up to 60%. While this requires upfront investment in marketing and assumes greater regulatory responsibility as the “importer of record,” it yields superior long-term enterprise value.
| Strategy | Typical Gross Margin | Market Entry Speed | Distributor Responsibility |
|---|---|---|---|
| Branded Distribution | 20% – 30% | Fast (Pre-existing trust) | Low (Sales and logistics only) |
| OEM / Private Label | 40% – 60% | Moderate (Requires brand building) | High (Regulatory importer, marketing) |
Procurement decision criteria
When executing the final sourcing decision, procurement criteria must be strictly weighted. Unit cost is critical, but it must never compromise the maximum acceptable defect rate, which should be aggressively targeted at less than 0.5% for structural failures.
Furthermore, buyers should prioritize suppliers capable of providing a comprehensive suite of orthodontic products, ensuring a streamlined supply chain and a unified standard of quality across
Key Takeaways
- Wholesale sourcing and supply-chain implications for orthodontic buccal tube
- Specifications, compliance, and commercial terms buyers should validate
- Actionable recommendations for distributors and procurement teams
Frequently Asked Questions
What does an orthodontic buccal tube do?
It anchors the archwire on molars and helps deliver force for tooth movement, rotation, and torque in fixed braces treatment.
Which slot sizes are most commonly used for buccal tubes?
The standard sizes are 0.018 inch and 0.022 inch. Clinics and distributors usually stock both to match different treatment systems.
Why is 17-4 stainless steel important in buccal tubes?
17-4 stainless steel offers high strength, corrosion resistance, and durability, helping reduce slot deformation and bond failure under heavy molar forces.
Should I choose bondable or weldable buccal tubes?
Choose bondable tubes for direct enamel bonding and weldable tubes for use with molar bands. The best option depends on your clinical workflow and case preference.
Why do buyers choose Denrotary buccal tubes?
Denrotary offers strong-bond monoblock buccal tubes within a full orthodontic portfolio, backed by medical-grade manufacturing and CE, FDA, and ISO13485 certifications.
Post time: Apr-17-2026