Orthodontic Market Trends Driving B2B Demand
The global orthodontic supplies market is experiencing sustained expansion, driven by an increasing clinical focus on precise tooth movement and growing patient demand for aesthetic dental solutions. For B2B wholesale buyers and distributors, this translates into a rapidly scaling procurement environment. Industry analyses project the market to maintain a compound annual growth rate (CAGR) of approximately 8.5% over the next five years, pushing total valuation well beyond previous thresholds. This growth necessitates a strategic realignment of supply chains; distributors must secure reliable, high-volume inventory to meet the escalating requirements of dental clinics and orthodontic networks.
Key demand drivers for wholesale buyers
The primary catalyst for wholesale demand is the demographic expansion of orthodontic patients, particularly the surge in adult orthodontics. Adults now account for nearly 30% of all orthodontic patients globally, fundamentally altering the product mix required by clinics. This demographic prioritizes reduced chair time and low-visibility appliances, directly driving the bulk procurement of self-ligating systems, aesthetic ceramic brackets, and clear sapphire options.
Additionally, rising disposable incomes in emerging economies have democratized access to advanced dental care. For procurement teams, this means that while premium markets demand advanced low-friction brackets, there remains a massive, volume-driven requirement for standard metal brackets and traditional archwires in developing regions.
Distributor opportunities in growing orthodontic markets
Distributors possess significant opportunities to capture market share by targeting high-growth regions with optimized product portfolios. By aligning with manufacturers capable of sustaining a weekly output of 10,000 to 15,000 bracket sets, distributors can ensure uninterrupted supply to expanding clinical networks.
Furthermore, the transition toward self-ligating brackets offers a lucrative avenue for B2B channels. Because these systems reduce clinical adjustment times and eliminate the need for elastomeric ligatures, clinics are increasingly willing to commit to higher-volume annual purchasing contracts. Distributors who secure exclusive regional rights to high-quality, medical-grade self-ligating portfolios can command strong competitive advantages and higher retention rates among their clinical clientele.
Product Mix Trends for Orthodontic Buyers
Optimizing the product mix is critical for orthodontic distributors aiming to service diverse clinical needs. A comprehensive portfolio must balance traditional, cost-effective mechanical systems with advanced, aesthetically pleasing alternatives. The modern orthodontic catalog spans complex brackets to essential consumables, each requiring specific technical evaluations during the sourcing process.
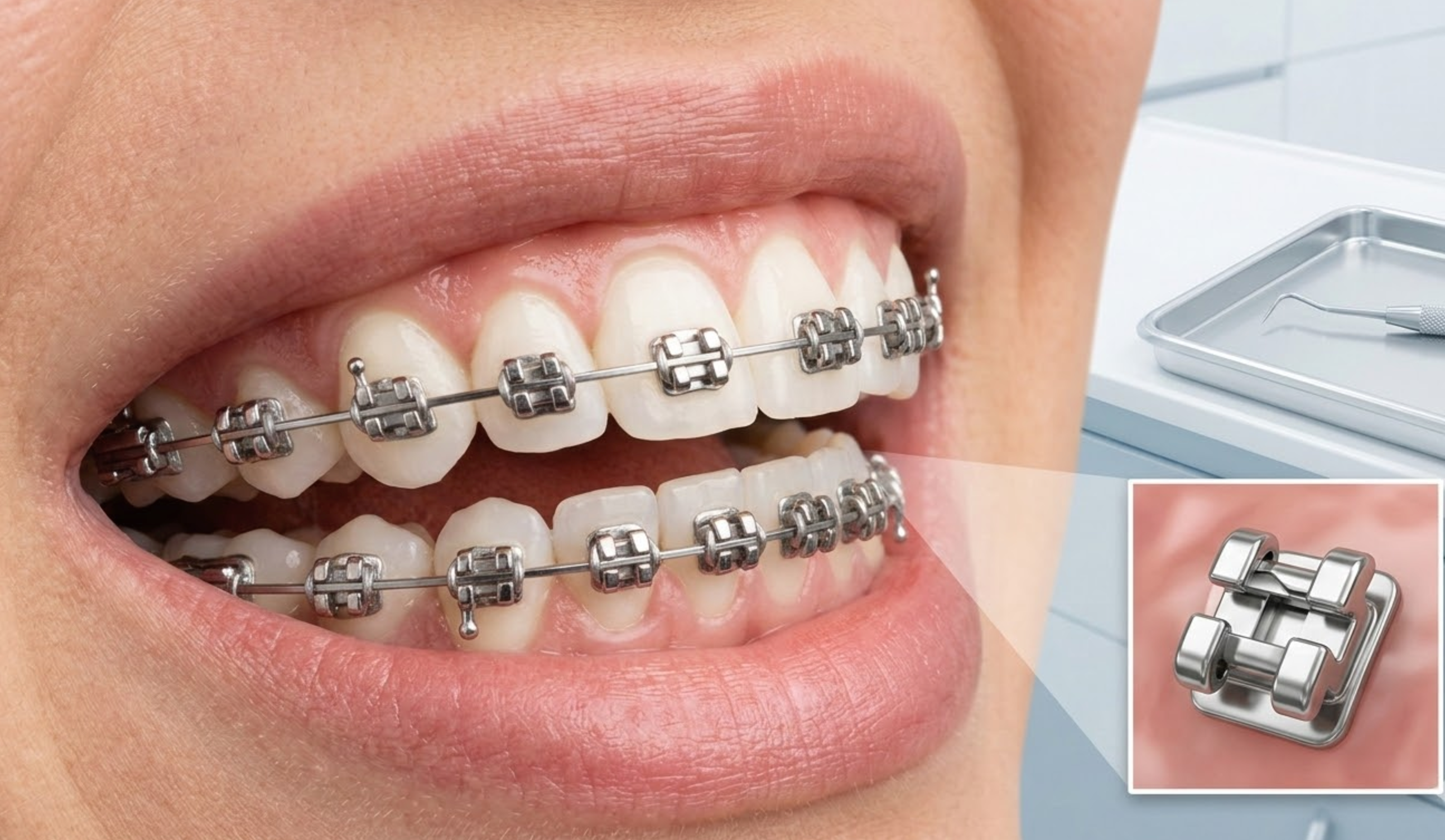
Metal, ceramic, sapphire, and self-ligating brackets
The foundational component of any orthodontic portfolio is the bracket system. Leading manufacturers utilize Metal Injection Molding (MIM) technology with 17-4 stainless steel to produce brackets with high tensile strength and precise slot dimensions. This manufacturing standard minimizes friction and enhances the efficiency of tooth movement.
Beyond standard metal, the demand for aesthetic options requires distributors to stock ceramic and sapphire brackets. Ceramic brackets offer a translucent appearance but require careful manufacturing to prevent brittleness, while monocrystalline sapphire brackets provide superior clarity and stain resistance. Self-ligating brackets, featuring integrated sliding mechanisms, represent the fastest-growing segment due to their ability to lower friction and reduce treatment duration by several months.
Archwires, buccal tubes, elastics, and accessories
A robust B2B catalog must also encompass the full spectrum of auxiliary components. Archwires, primarily manufactured from Nickel-Titanium (NiTi) and stainless steel, are essential for applying continuous force. Distributors should ensure their suppliers provide wires with consistent transition temperatures and shape memory.
Other critical accessories include strong-bond monoblock buccal tubes, which offer superior adhesion and patient comfort compared to traditional welded bands. For elastomeric products like power chains and ligature ties, procurement teams must source latex-free polyurethane materials that guarantee high stretch, excellent rebound memory, and resistance to clinical staining.
Product comparison criteria for bracket and wire selection
When comparing products for wholesale selection, buyers must evaluate material composition, dimensional tolerance, and mechanical reliability. Below is a comparative overview of primary bracket materials to guide portfolio diversification:
| Bracket Material | Manufacturing Process | Key Advantage | Target Clinical Segment |
|---|---|---|---|
| 17-4 Stainless Steel | Metal Injection Molding (MIM) | Maximum durability, low friction | Traditional & pediatric cases |
| Ceramic (Polycrystalline) | Sintering / Milling | Tooth-colored aesthetics | Adult aesthetic cases |
| Sapphire (Monocrystalline) | Diamond Milled / Heat Treated | Total transparency, no staining | Premium adult aesthetics |
| Self-Ligating (Metal/Clear) | MIM + Precision Assembly | Reduced chair time, sliding mechanics | High-efficiency modern clinics |
B2B Sourcing Strategy for Orthodontic Products
Establishing a resilient B2B sourcing strategy requires more than identifying competitive pricing; it demands rigorous supplier vetting and scalable procurement frameworks. As the orthodontic market expands, buyers must mitigate supply chain vulnerabilities by partnering with manufacturers who demonstrate robust technical capabilities and flexible commercial terms.
Supplier capabilities for stable quality and scale
Stable quality at scale is the hallmark of a tier-one orthodontic manufacturer. Procurement teams should prioritize suppliers operating modern, medical-grade workshops equipped with advanced machinery, such as precision German milling and molding equipment. Facilities utilizing automated bracket production lines offer superior consistency, effectively eliminating the micro-variations that plague manual assembly.
A supplier’s production capacity is a direct indicator of their ability to support a distributor’s growth. Manufacturers capable of producing upwards of 10,000 pieces weekly demonstrate the operational bandwidth necessary to fulfill large-scale, recurring B2B orders without extending lead times during peak demand cycles.
MOQ, private label, packaging, and customization options
Commercial flexibility is essential for distributors building proprietary brands. Standard minimum order quantities (MOQs) for orthodontic brackets typically range from 500 to 1,000 sets for OEM or private label production. Buyers should negotiate these thresholds based on their inventory turnover rates.
Customization extends beyond branding to include packaging configurations. Suppliers should offer tailored packaging solutions, such as individual patient kits or bulk clinical dispensers, accompanied by localized labeling. Private labeling allows distributors to build regional brand equity while leveraging the manufacturer’s established quality control infrastructure.
Procurement steps to reduce sourcing risk
To reduce sourcing risk, B2B buyers must implement a phased procurement methodology. This begins with comprehensive factory audits to verify manufacturing equipment and cleanroom standards. Following the audit, buyers should request pre-production samples to test bracket slot tolerances and buccal tube bond strength.
Once samples are approved, initializing a trial order of 100 to 200 sets allows the distributor to evaluate the supplier’s logistics, packaging integrity, and adherence to lead times before committing to full-scale annual supply contracts. This step-by-step validation minimizes financial exposure and ensures clinical reliability.
Compliance, Quality, and Logistics Requirements
Orthodontic supplies are classified as medical devices, necessitating strict adherence to international regulatory frameworks. B2B buyers cannot compromise on compliance, as importing non-compliant products risks customs seizures, legal liabilities, and severe damage to a distributor’s reputation among clinical end-users.
Certifications, standards, and required documentation
Procurement teams must ensure that suppliers possess the requisite certifications for the target market. At a minimum, this includes ISO 13485 certification, which validates the manufacturer’s quality management system specifically for medical devices. For distribution in the European Union, a valid CE mark is mandatory, while entry into the United States requires FDA registration and appropriate 510(k) clearances for applicable devices.
Buyers must request and independently verify the supplier’s Declaration of Conformity and detailed technical files. Maintaining an updated repository of these compliance documents is critical for smooth customs clearance and downstream clinical audits.
Packaging, shelf life, labeling, and shipment readiness
Proper packaging and labeling are heavily regulated aspects of orthodontic logistics. Products must feature Unique Device Identification (UDI) barcodes to ensure full traceability from the factory floor to the patient. Shelf life is a particularly critical metric for elastomeric products like power chains and elastics, which typically have an optimal performance window of 18 to 24 months.
Logistics readiness also involves environmental controls. Polyurethane-based accessories are sensitive to extreme temperatures and UV exposure. Distributors must mandate climate-controlled shipping and opaque, sealed packaging to prevent material degradation during international transit.
Quality inspection checkpoints for orthodontic products
Implementing strict quality inspection checkpoints is non-negotiable. B2B buyers should establish acceptable quality limits (AQL) with their suppliers, typically demanding a defect rate of less than 0.5% for high-volume bracket orders. Dimensional accuracy is paramount; slot tolerances must generally be maintained within ±0.015mm to ensure proper archwire engagement.
| Inspection Parameter | Target Standard / Tolerance | Verification Method |
|---|---|---|
| Bracket Slot Dimensions | ±0.015mm | Calibrated optical comparator |
| MIM Base Integrity | Zero voids, 80-gauge mesh equivalent | Microscopic visual inspection |
| Elastomeric Rebound | >90% recovery after 24h stretch | Tensile stress testing |
| Packaging Seal | Complete hermetic seal | Vacuum leak test |
Turning Market Trends into Procurement Margin
Translating global orthodontic trends into tangible procurement margins requires a sophisticated approach to portfolio management and pricing architecture. Wholesale buyers must leverage their sourcing efficiencies to offer competitive pricing to clinics while protecting their own profitability metrics.
Pricing, margin, and bundling models
Effective pricing models in the B2B orthodontic space often rely on strategic bundling. By combining high-margin aesthetic brackets or self-ligating systems with high-volume, lower-margin consumables like archwires and buccal tubes, distributors can create comprehensive ‘patient start kits’.
Wholesale margins on individual consumable components typically range from 35% to 55%, depending on the sourcing volume. However, by transitioning clinics to bundled purchasing models, distributors can lock in recurring revenue streams, reduce aggregate shipping costs, and elevate the overall gross margin of the account.
Supplier and portfolio selection framework
Selecting the right supplier portfolio involves balancing premium innovations with reliable commodities. A profitable framework requires sourcing standard 17-4 stainless steel brackets from high-capacity manufacturers to capture volume-driven, cost-conscious clinics. Simultaneously, the distributor must secure specialized lines—such as low-friction self-ligating systems or monocrystalline sapphire brackets—to penetrate premium orthodontic practices.
Consolidating these distinct product lines under one or two highly capable manufacturers reduces freight overhead and simplifies regulatory compliance, directly impacting the bottom line.
Decision checklist for profitable orthodontic sourcing
To ensure profitable sourcing, procurement teams should utilize a standardized decision checklist. First, verify the supplier’s volume capacity (e.g., >10,000 sets/week) to guarantee scale. Second, validate ISO 13485, CE, and FDA compliance to eliminate market entry barriers. Third, assess the availability of private label customization at manageable MOQs.
Finally, conduct a total landed cost analysis that factors in unit price, climate-controlled freight for sensitive elastomers, and import duties. By systematically checking these parameters, B2B buyers can confidently transform emerging orthodontic market demands into sustainable, high-margin revenue streams.
Key Takeaways
- Wholesale sourcing and supply-chain implications for Orthodontic Market Trends
- Specifications, compliance, and commercial terms buyers should validate
- Actionable recommendations for distributors and procurement teams
Frequently Asked Questions
What products are seeing the fastest growth in the orthodontic market?
Self-ligating, ceramic, and sapphire brackets are growing fastest, driven by adult demand for lower visibility and shorter chair time.
Why are self-ligating brackets gaining more B2B demand?
They reduce friction, shorten adjustment visits, and can cut treatment time, making clinics more willing to place larger repeat orders.
How should distributors balance bracket portfolios across regions?
Stock premium aesthetic systems for mature markets and dependable metal brackets plus standard archwires for volume-led emerging markets.
What should buyers check when sourcing archwires and accessories?
Verify consistent wire properties, strong-bond monoblock buccal tubes, and latex-free power chains with good stretch, rebound, and stain resistance.
Why consider Denrotary for orthodontic wholesale supply?
Denrotary offers brackets, wires, and accessories with CE, FDA, and ISO13485 standards, plus scalable production and medical-grade manufacturing.
Post time: Apr-20-2026